Medical Translation Services in India
Trusted by
![Healthcare Translation Services 1 airbu[1]](https://anantranslations.com/wp-content/uploads/2026/02/airbu1-1-e1771225257160.avif)




Specialized Medical Translation Solutions
We provide professional document translation services for critical healthcare and regulatory materials, including:
- Medical records
- Medical reports
- Doctor Prescriptions
- Discharge summaries
- Case study documentation
- Diagnostic reports
- Insurance documentation
- Technical documents
Our professional medical translators ensure terminology consistency using translation memories (TMs) and glossary control to maintain accuracy across large projects.
Need to bulk translate medical reports? Our scalable workflow allows our translators to work efficiently on high-volume assignments without compromising quality.
We offer specialized clinical trial document translation in India for global regulatory bodies and European trials.
Services include:
- Informed Consent Form (ICF) translation services for Europe
- Clinical Study Protocols
- Investigator Brochures
- Case Report Forms (CRFs)
- Clinical Outcome Assessments (COAs)
- Safety narratives
Our localization service ensures accurate translation content adapted to local regulations and patient comprehension standards. We provide CRO clinical trial translation support with timely delivery and regulatory formatting compliance.
As a professional pharma regulatory translation services provider in India, we assist with:
- Pharmaceutical dossier translation services
- CTD translation services (Common Technical Document)
- SmPC translation
- Regulatory submissions
- Stability reports
- Drug safety documentation
Our translators work closely with subject experts to ensure compliance with EMA and EU regulatory standards. All translation content is prepared for submission to regulatory bodies without delays.
We support medical device manufacturers with EU MDR medical document translation, including:
- Medical device IFU translation services
- Technical documentation
- Risk management files
- Clinical evaluation reports
- Patient leaflet translation services in India
- CE marking documentation
Our document translators ensure strict control of terminology and compliance with EU MDR guidelines while maintaining clarity in the target language.
Planning global expansion?
We provide multilingual medical language translation for export markets, including:
- European language medical translation (French, German, Spanish, Italian, Dutch, Polish)
- Arabic medical translation
- Russian medical translation
- Asian language support
Our professional translators combine medical expertise with cultural sensitivity to ensure accurate communication across markets.
Industries We Serve
Specialized medical translation solutions tailored to regulated healthcare sectors.
Certified Medical & Clinical Trial Translation for Global Regulatory Compliance
At Anan Translations our team of professional translators delivers certified medical and clinical trial translation services for pharmaceutical companies, CROs, medical device manufacturers, hospitals, and exporters.
As one of the reliable translation companies serving the healthcare sector, we ensure your documents are accurately translated from the source language into your required target language while meeting all regulatory requirements for official submission.
We help organizations translate documents for EU submissions, clinical trials, regulatory approvals, and global market entry — with full subject-matter expertise and cultural sensitivity.
Medical Subject-Matter Experts
Native Linguists
On-Time Delivery
Trusted Medical Translation Company for Regulated Industries
As a specialized medical translation company for clinical trials in India, we support:
- Pharmaceutical manufacturers
- Contract Research Organizations (CROs)
- Medical device companies
- Biotechnology firms
- Regulatory consultants
- Healthcare exporters
If you require translating documents for EU submissions or large-scale projects, Anan Translations can manage bulk medical translation assignments with structured workflows and dedicated project managers.

Why Choose Anan Translations?
A specialized medical translation partner for regulated industries.
Typical Translation Providers
General Translators
Lack subject-matter expertise in healthcare and life sciences.
Limited Regulatory Knowledge
Minimal alignment with EU, EMA, MDR, or global regulatory bodies.
Basic Translation Tools
No structured terminology databases or translation memory control.
No Dedicated Coordination
Projects handled without centralized project management.
Non-Certified Output
Translations may not meet official or regulatory submission standards.
Limited Scalability
Struggle with large-scale or multi-country medical projects.
Anan Translations
Professional Medical Translators
Our translators have deep subject-matter expertise in medicine, pharmacy, biotechnology, and life sciences.
Regulatory Compliance Focus
We align translation projects with EU, EMA, MDR, and other global regulatory bodies.
Advanced Translation Technology
We use translation memories and terminology databases to ensure consistency across technical documents and ongoing projects.
Dedicated Project Managers
Experienced project managers coordinate timelines, quality checks, and client communication for smooth execution.
Certified Document Translation Services
Certified medical translation services in India for official and regulatory use.
Bulk Medical Translation Capability
We handle large-scale assignments with structured workflows and reliable delivery timelines.
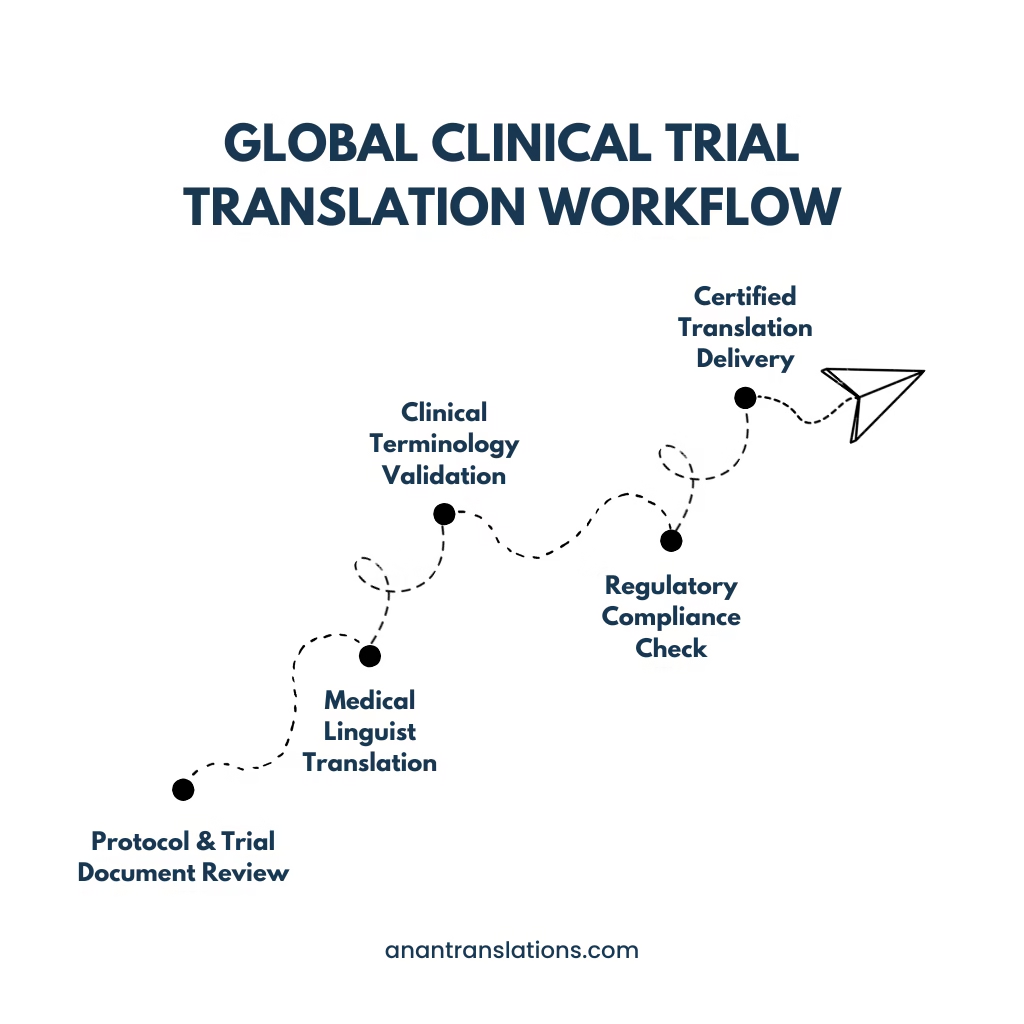
Our Healthcare Translation Process
Ensuring medical accuracy, patient safety, and regulatory compliance in every translation.
Medical Content & Terminology Analysis
We carefully review your medical documents and analyze complex terminology before translation begins.
Translation by Healthcare Subject-Matter Experts
Certified translators with healthcare expertise handle your documents for precise and accurate translation.
Bilingual Medical & Linguistic Review
A second set of bilingual reviewers ensures linguistic accuracy and adherence to medical standards.
Multi-Step Quality Assurance & Consistency Checks
Multiple QA stages check for consistency, terminology accuracy, and overall translation quality.
Final Validation for Regulatory & Patient Safety Compliance
Every translation is verified for regulatory compliance and patient safety before delivery.
Bulk Medical Translation for Pharma & CROs
Managing multiple clinical trials or EU submissions? We provide:
- Dedicated account management
- Volume-based pricing
- Structured delivery timelines
- Support for multi-country submissions
- CTD dossier translation
- Ongoing regulatory translation services

